Sensor Module
The heart of ChemDAQ monitoring systems, the ChemDAQ sensor modules feature excellent performance as well as ChemDAQ’s proprietary Spot-On™ filter technology to reduce cross interference and precisely measure gas vapor levels where applicable (e.g. PAA and EtO). The sensors come in two main configurations, the Steri-Trac® sensor module and the E-Cell®
sensor module.

E-Cell Sensor
ChemDAQ’s E-Cell sensor is used to precisely measure gas/vapor levels for PAA and H2O2. Unlike other suppliers, ChemDAQ’s E-Cell sensors are calibrated with PAA and H2O2 test gas to ensure top safety and performance.

Steri-Trac® Sensor
ChemDAQ’s Steri-Trac Sensor is used to monitor EtO, Oxygen (O2) and Lower Explosive Limit (LEL) and is available for intrinsically safe applications. The Steri-Trac sensors communicate via RS-485 output and so can be used over long distances.
Portable Monitoring
SafeCide™ Series
Quickly and easily detect hazardous areas the SafeCide series, a portable monitoring system to provide real-time measurements on the floor and easily identify hot spots and leaks.
Fixed Monitoring
Steri-Trac® Series
ChemDAQ’s premier fixed area monitoring system, the Steri-Trac series of products are designed to continuously measure and alert you to hazardous vapor levels in the air.
Sensor Communication Interface (SCI™)
The SCI is a multi-configurable monitoring solution to accurately and reliably detect vapors throughout your facility and communicate readings to a compatible data system for enhanced connectivity.
VaporAlert™ Series
The first and only water-resistant monitoring system on the market, the VaporAlert is designed to continuously monitor in the wet areas of your plant.
Compatible Products

Data Acquisition System (DAQ®)
ChemDAQ’s DAQ is an area monitoring and archival computer software solution. The DAQ can sync and centralize data from up to 32 monitoring locations simultaneously; compare all exposure data to occupational exposure limits; and provide historic reporting for each monitoring point.

Remote Display
Remote displays added to ChemDAQ monitors increase safety by warning employees of hazardous conditions before they enter the area. Display a single reading from a specific space on this easy-to-install display.
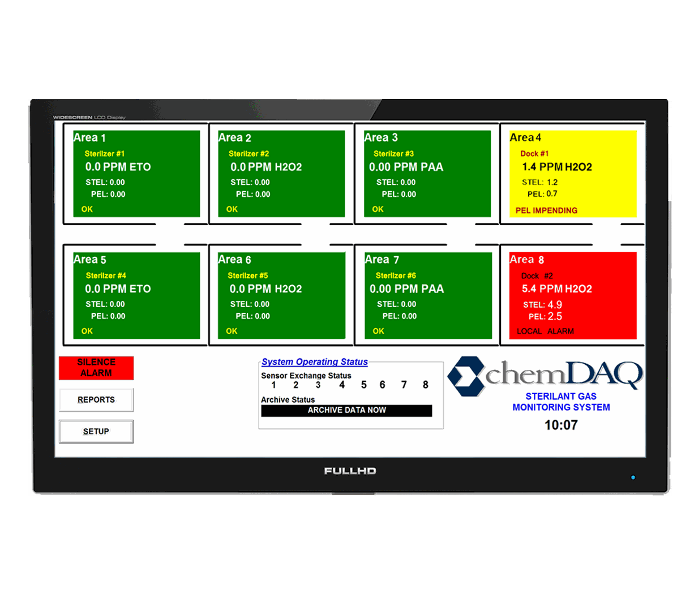
Remote Video Display
Remote video displays are an extension of your DAQ. The user can choose to highlight certain areas from their DAQ to display on this single, easy-to-read monitor.
Connect with a Product Expert
Need help determining which monitoring system best fits your needs? Connect with a product expert.




